Ozone Room Sterilisation Unit
Ozone Room Sterilisation UnitThe Principle Function:
Ozone is the fastest-acting oxidising disinfectant and the most efficient broad-spectrum, microbiological control agent commercially available. Being highly reactive and completely non-discriminating, it immediately starts oxidising anything it comes in contact with. This makes ozone a very powerful bactericide agent.
Ozone is less stable than oxygen. When ozone comes in contact with bacteria, an oxygen atom breaks away from the ozone molecule, oxidising and thereby neutralising the bacteria.
Unlike any other oxidising agent, ozone decomposes into environmentally friendly oxygen, leaving no toxic residues.
What is Ozone and how it is produced?
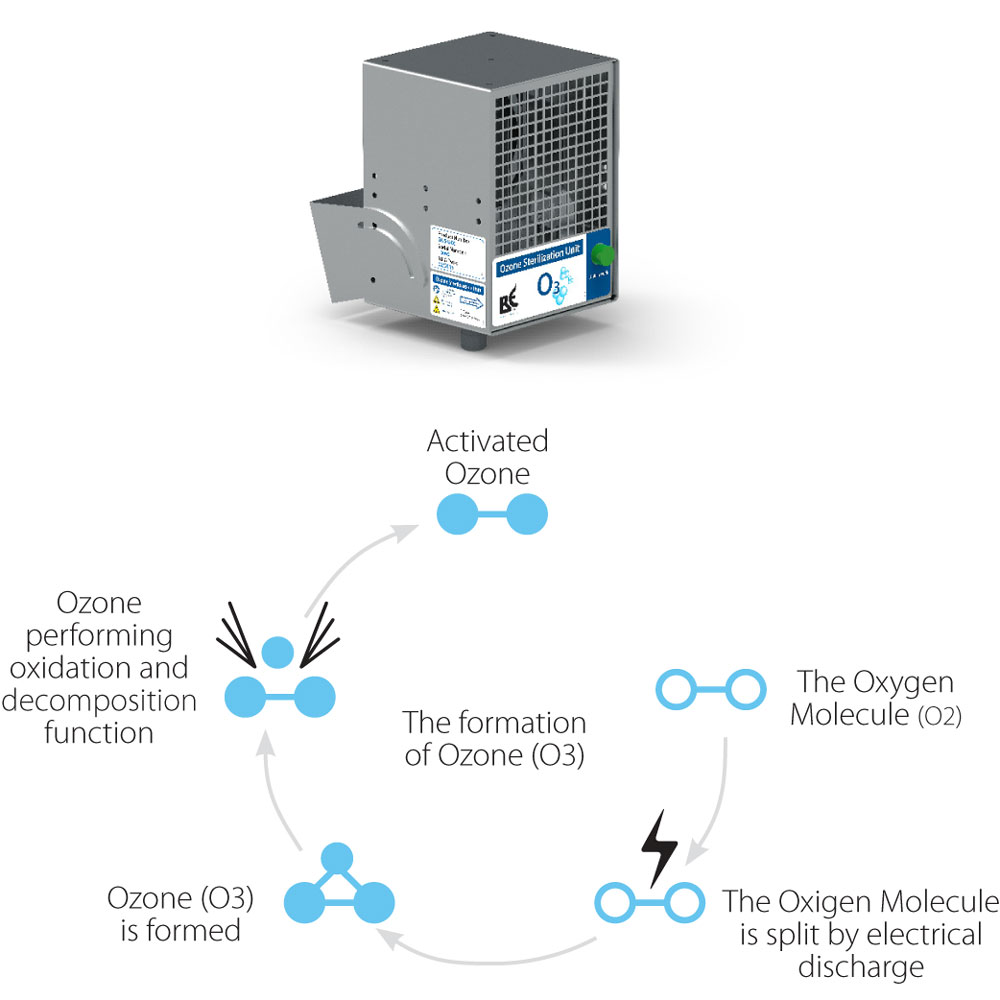
Ozone, or trioxygen, is a fast-acting oxidising disinfectant made up of three oxygen atoms, O3. Ozone occurs naturally in the upper atmosphere, or stratosphere, when the sun’s light splits oxygen molecules, O2, into separate atoms. When these single atoms bond with other oxygen molecules, they form ozone, protecting us from the sun’s harmful ultraviolet rays.
Commercially, ozone can be used to disinfect, deodorise, and sanitise the air, sterilise equipment, and remediate mold. Ozone also kills microorganisms, such as bacteria, in drinking water, pools, and spas.
Where to use the Ozone Room Sterilisation Unit?
- Disinfect air in operating rooms, doctors’ offices, and healthcare facilities
- Deodorise clothing and fabrics from smoke or pet smells
- Kill bacteria on food and food packaging areas
- Remove mold spores and yeast from the air for food processing
- Kill insects in storage areas
OZONE Production
The AORT system contain a mechanical timer that enables you to set in advance the ending time of the Ozone sterilization process.
AORS system

The AOR Stand enables the combination of up to four AOR systems, in a flexible and highly mobile adjustment for a maximum cover of the designated space for sterilization.
 English
English Русский
Русский


